News
The Bonlab goes DIY with a microfluidic device for fabrication of double emulsion droplets and polymer microcapsules
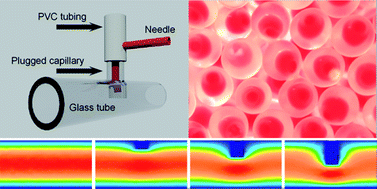
The Bonlab lead by assoc. prof. Stefan Bon publishes in Polymer Chemistry how to make a DIY microfluidic device to fabricate droplets-in-droplets. They demonstrate that by using a syringe needle, plastic tubing, two glass capillaries and epoxy glue a microfluidic device can be fabricated straightforwardly that allows for the production of double emulsions, or in other words the generation of droplets-in-droplets. The device in essence is a serial combination of droplet generation by co-flow and a T-junction. To reduce potential issues with channel wetting, we established that an “obstructed” T-junction outperformed a conventional T-junction. They illustrate the versatility of our device through production of a range of polymer microcapsules, including ones that contain a waterborne dispersion of colour changing pigment, and microcapsules with compartmentalized ferrofluidic segments, that is capsules that contain more than one droplet of ferrofluid.
To read the paper:
To find out more about the BonLab: , Twitter: , Youtube:
Quadruple hydrogen bond polymer colloids HIPE up gels
Scientists in the team of assoc. prof. Stefan Bon (BonLab) have developed a convenient route to organogels templated by high internal phase emulsions (HIPEs) of water droplets which they refer to as HIPE- gels. Key is the use of a waterborne polymer latex loaded with a multiple hydrogen bond (MHB) array functionality - here 2-ureido-4[1H] pyrimidinone (UPy). Upon contact with an organic phase is a good solvent for the polymer (by shaking), swelling, and subsequent partial disentanglement of polymer chains originating from and wrapped up in the form of a latex particle, and phase transfer of polymer into the organic phase occurs. This process promotes the formation of a cross-linked swollen network in the form of an organogel through quadruple hydrogen bond interactions. Typically a HIPE-gel formulation contains 85vol% dispersed water droplets in 15vol% toluene at an original loading of 0.1wt% of UPy-functionalized latex particles in the water phase. They demonstrate that the HIPE-gels can be used as a soft matter storage and slow release material. The closed-cell compartmentalized water droplets remain intact upon submersion of the HIPE-gel into water, even under high osmotic pressure gradients. Upon drying the HIPE-gel a self-supporting porous monolith is obtained. They believe that the approach to the fabrication of the HIPE gels opens routes for potential applications in medical injectable gels, pressure sensors, and other interesting multiphase soft matter materials.
The work is published in chemical communications,
More on the BonLab,
‘Left-handed iron corkscrews’ point the way to new weapon in battle against superbugs like MRSA

 Scientists at the University of ����TV have taken inspiration from corkscrew structures found in nature to develop a new weapon in the fight against infections like E-coli and MRSA.
Scientists at the University of ����TV have taken inspiration from corkscrew structures found in nature to develop a new weapon in the fight against infections like E-coli and MRSA.
Researchers have created a new synthetic class of helix-shaped molecules which they believe could be a key tool in the worldwide battle against antibiotic resistance. By twisting molecules around iron atoms they have created what they term ‘flexicates’ which are active against MRSA and E-coli - but which also appear to have low toxicity , reducing the potential for side effects if used in treatment. The work is published in Nature Chemistry.
The new structures harness the phenomenon of ‘chirality’ or ‘handedness’ whereby the corkscrew molecules could be left-handed or right-handed. By making the most effective ‘hand’ to attack a specific disease, the University of ����TV research paves the way towards a more targeted approach to killing pathogens. In the case of E-coli and MRSA, it is the left ‘hand’ which is most effective.
Professor Peter Scott of the University of ����TV’s chemistry department said although this particular study concentrated on flexicates’ activity against MRSA and E-coli, the new method of assembly could also result in new treatments for other diseases.
“It’s a whole new area of chemistry that really opens up the landscape to other practical uses. These new molecules are synthetically flexible, which means that with a bit of tweaking they can be put to use against a whole host of different diseases, not just bugs like MRSA which are rapidly developing resistance to traditional antibiotics. Flexicates are also easier to make and produce less waste than many current antibiotics.”
Scientists have long been able to copy nature’s corkscrew-shaped molecules in man-made structures known as helicates – but they have thus far not been able to use them in fighting diseases. One of the key issues is the problem of handedness. Sometimes ‘left-handed’ molecules in drugs are the most effective at combating some disease, while sometimes the ‘right-handed’ version works best. Until now, scientists working with helicates have found it difficult to make samples containing just one type of corkscrew; either the right- or left-handed twist.
With flexicates, the University of ����TV scientists have succeeded in making samples containing just one type of twist – resulting in a more targeted approach which would allow the drug dosage to be halved. Flexicates solve other problems encountered by helicates, as they are easier to optimise for specific purposes, are better absorbed by the body and are easier to mass-produce synthetically.
Professor Scott said: “Drugs often have this property of handedness - their molecules can exist in both right and left handed versions but the body prefers to use only one of them. For this reason, drug companies have to go to the trouble of making many traditional molecules as one hand only. What we have done is solve the ‘handedness’ problem for this new type of drug molecule. By getting the correct hand we can halve the drug dose, which has the benefits of minimising side effects and reducing waste. For patients, it’s safer to swallow half the amount of a drug. Our work means that we can now make whichever hand of the corkscrew we want, depending on the job we require it to do.”
Notes to editors
The study, entitled Optically pure, water-stable metallo-helical ‘flexicate’ assemblies with antibiotic activity, is published in Nature Chemistry.
The research was also supported financially by EPSRC.
It is authored by Peter Scott, Suzanne Howson, Guy Clarkson and Alison Rodger from the University of ����TV, Albert Bolhuis from the University of Bath and Viktor Brabec and Jaroslav Malina from the Academy of Sciences of the Czech Republic.
When the paper is published it can be retrieved at
Contact details
Professor Peter Scott is available on +44 (0) 24 7652 3238 or peter.scott@warwick.ac.uk
University of ����TV press officer Anna Blackaby is available on + 44 (0) 2476 575910 or + 44 (0)7785 433155 or a.blackaby@warwick.ac.uk
Front Page Artwork by Elisabeth Heissler
Precious metal materials
, in collaboration with colleagues in the Department of Physics and at Johnson Matthey plc have a paper published this week in the RSC journal Chemical Science: this describes mild synthetic chemical routes to complex extended structures that contain the metal iridium in various oxidation states. This illustrates the scope for the discovery of new functional materials by exploration of novel reaction conditions and using the chemistry of lesser studied elements.
Prof. Patrick Unwin made an ISE fellow
Professor Patrick Unwin, Department of Chemistry has been made a Fellow of the International Society of Electrochemistry (ISE). This award comes in recognition of the outstanding contributions Professor Unwin has made to the field of electrochemistry throughout his career.
Professor Unwin leads the ����TV Electrochemistry and Interfaces Group, who are known for developing innovative methods for functional imaging of surfaces and interfaces. They are currently in the process of developing a whole suite of new nanoscale electrochemical imaging methods with support from the European Research Council.
Professor Unwin said:
"I have been fortunate to have many good colleagues and fantastic students and postdocs in my 20 years at ����TV. They are great fun to work with and have contributed significantly to me winning this award."
About ISE
ISE is a large non-profit-making organisation set up to promote work done in the field of electrochemistry and provide an international community for those involved. It has around 2,300 members based in more than 60 countries worldwide.
Prof. David Haddleton receives Royal Society Wolfson Research Merit Award
|
Professor David Haddleton, Department of Chemistry, has been awarded a Wolfson Research Merit Award by the Royal Society, the UK's national academy of science. This award aims to provide universities with additional support to attract to this country and retain respected scientists of outstanding achievement and potential. It is jointly funded by the Wolfson Foundation and the Department for ����TV, Innovation and Skills. Professor Haddleton will be working on a project entitled "Controlled polymerisation to new materials: Polymer therapeutics to oil recovery". He will take up his award by 1 May 2011. (see also ) |
University of ����TV & partners awarded £1.4 million in national solar energy programme
The University of ����TV is a key part of a consortium that has just been awarded £1.4 Million by a new national programme designed to develop the next generation of solar energy harvesting technology.
The UK’s Technology Strategy Board and the Engineering and Physical Sciences Research Council has awarded the £1.4 million to a consortium that includes The University of ����TV; the companies Kurt Lesker, Asylum Research, New World Solar, and Molecular Solar, and Imperial College London. Together they will work on the development of Prototype High Efficiency Multi-Junction Organic Solar Cells.
Professor Tim Jones from the University of ����TV said:
“We are working with solar cells made from organic semiconductor materials which offer the prospect of very low cost manufacture of lightweight, flexible cells. They are made from sustainable materials and can be deployed as flexible sheets that could be used for a variety of applications including: a solar powered mobile phone charger that’s rolls up into a shape as small as the size of a pen, micro-lights that can be added to clothing, and a detachable sun-shade for automobile windscreens that powers a small integral fan to circulate air and cool the interior of the car when parked in direct sunlight.”
Peter Ballantyne from Molecular Solar, a spin-out company from the University of ����TV, which will be developing this new technology said:
“The low cost and flexibility of this new technology will lead to new applications that will further accelerate the growth of the solar power market, which has seen 40%/year growth over the last 10 years. Just one significant opportunity in consumer applications is the area of mobile phone chargers where over 1.3 billion units a year are produced.”
In total fifteen British businesses and seven universities will share £5 million of government funding from the UK’s Technology Strategy Board and the Engineering and Physical Sciences Research Council to enable them to research the use of novel nanoscale technologies to develop the next generation of solar energy harvesting.
Iain Gray, Chief Executive of the Technology Strategy Board said:
“These projects will help to position British businesses to exploit the growing global demand for solar energy harvesting technologies – and in the process help grow the British economy – while at the same time provide sustainable energy solutions for the UK. The projects are great examples of how to transfer commercially-focused research into the business community.”
David Delpy, Chief Executive of the Engineering and Physical Sciences Research Council said:
“This is the first example of Nanoscience research funding from the Research Councils being directly pulled through to application funding with the Technology Strategy Board via a stage-gated funding route. This approach actively supports economic growth whilst helping to solve one of society's greatest challenges.”
For further information please contact:
Professor Tm Jones University of ����TV Tel: +44 (0)2476 528265
Email: 

 t.s.jones@warwick.ac.uk
t.s.jones@warwick.ac.uk
Peter Dunn, Head of Communications
Communications Office, University House,
University of ����TV, Coventry, CV4 8UW, United Kingdom
email: p.j.dunn@warwick.ac.uk

Tel: +44 (0)24 76 523708 Mobile/Cell: +44 (0)7767 655860
PR34 25th March 2011
Bon Lab features on the cover of Polymer Chemistry
 |
A paper by PhD student Nicholas Ballard and associate professor Stefan Bon, previously selected by the Royal Society of Chemistry as a hot paper, features on the April 2011 cover of Polymer Chemistry. The paper entitled Hybrid biological spores wrapped in a mesh composed of interpenetrating polymer nanoparticles as "patchy" Pickering stabilizers describes a new method for the decoration of the intricate morphology of spore particles with polymer nanoparticles and investigates their behaviour at liquid–liquid interfaces. It is found that a large difference in the interfacial activity between spherical microspheres and the anisotropic particles synthesized exists, which is explained in terms of particle wettability. ; more on the |
Graphene oxide’s solubility disappears in the wash
|
Drs Rourke and Wilson’s team made their discovery when treating the graphene oxide with sodium hydroxide (NaOH) in an attempt to increase the usefulness of the oxygen containing functional groups believed to be bound to the graphene. Unfortunately it seemed to make things worse rather than better. Indeed at high enough concentrations of NaOH Dr Rourke was left with a black suspension. The ����TV led researchers recalled that it had been shown that oxidation debris adheres to carbon nanotubes but the weak nature of the connection of this oxidation debris to the carbon nanotubes meant that a wash with a base can simply remove the oxidative debris. Experiments showed that in that particular case oxidative debris was found to make up almost a quarter of the mass of the “oxidized carbon nanotubes”. The researchers felt a similar process maybe happening in the Graphene Oxide they were studying. The results may also help explain the inordinately high levels of oxygen people were claiming to find in graphene oxide. Chemists were already struggling to identify enough plausible carbon to oxygen bonds to accommodate the amounts of oxygen believed to form part of graphene oxide.
The remaining liquid was also dried to give a white powder that the ����TV researchers showed contained the “oxidative debris” or OD; the OD was shown to be made up exclusively of small, low molecular weight compounds (i.e. less than 100 atoms). The graphene oxide recovered from washing process formed about 64% of the mass of the “graphene oxide” at the start of the process. The recovered OD or oxidative debris formed at least 30% of the weight of the mass of the original “graphene oxide”. Drs Rourke and Wilson’s team believe this shows that much of the oxygen that was believed to be closely bonded to the carbon in the graphene oxide was actually not bonded at all but simply lying on top of the graphene sheets, loosely connected to them as “oxidative debris”. This oxidative debris contained a large quantity of oxygen that simply came out in the wash when the graphene oxide was treated with sodium hydroxide. This creates a significant proble Drs Rourke and Wilson say “Our results suggest that models for the structure of graphene oxide need revisiting. These results have important implications for the synthesis and application of chemically modified graphene particularly where direct covalent functionalization of the graphene lattice is required.” The paper entitled: The Real Graphene Oxide Revealed: Stripping the Oxidative Debris from the Graphene-like Sheets by Dr. Jonathan P. Rourke, Priyanka A. Pandey, Joseph J. Moore, Matthew Bates, Neil R Wilson (all of the University of ����TV), and Dr Ian A. Kinloch, Prof. Robert J. Young (The University of Manchester), has just been published in Angewandte Chemie DOI: 10.1002/anie.201007520. Notes for editors: The researchers thank Dave Hammond for help with thermogravimetric analysis (TGA), Lijiang Song for help with mass spectrometry, and Ajay Shukla for help with X-ray photoelectron spectroscopy (XPS), the Midlands Physics Alliance Graduate School for a scholarship. The TEM, TGA, and XPS instruments as well as the mass spectrometer used in this research were purchased with support from Advantage West Midlands (part funded by the European Regional Development Fund) as part of the Science City programme. For further information please contact: Dr Jonathan P. Rourke Peter Dunn, Head of Communications PR23 8th March 2011 |
Inorganic Materials book series
The fifth volume, Energy Materials, co-edited by , with Duncan Bruce (York) and Dermot O’Hare (Oxford), has been published this week by Wiley.

JACS cover for Challis group
Prof. Greg Challis and his team together with Thomson and co-workers feature on this weeks cover of the Journal of the American Chemical Society. The absolute and relative stereochemistry of streptorubin B, a brightly colored prodiginine antibiotic, has been determined. Challis and co-workers utilized a mutant of Streptomyces coelicolor to conduct a mutasynthesis using enantioenriched deuterium-labeled biosynthetic precursors, while Thomson and co-workers developed an enantioselective total synthesis via a 10-membered pyrrolophane intermediate. See Challis and co-workers, p 1793, and Thomson and co-workers, p 1799.

Understanding Stimuli-Responsive Biomaterials
and collaborators investigate in detail the behaviour of stimuli-responsive polymer-protein conjugates in Polymer Chemistry. It is shown that at in vivo concentrations and when measured in blood rather than water, the behaviour of these materials deviates significantly from what is normally expected.
Konstantinos Bebis, Mathew W. Jones, David M. Haddleton and Matthew I. Gibson*. Polymer Chemistry, 2011,




